This deep sea fish is from the family Melanocetidae in the order Lophiiforms (anglerfish), commonly referred to as black seadevils. “Melanos” deriving from the Greek word for black and “cetus” meaning whale or sea monster. There is only one known genus of the family, Melanocetus, found in 1864 by German zoologist Albert Günther. Since then an additional 13 species have been found, with 6 currently recognized species. The two most well known of which are the Melanocetus johnsonii and Melonocetus murrayi (Pietsch, 2009).
This genus of fish lives in both the mesopelagic, or twilight zone, 200-1000 meters below the surface, and bathypelagic zone, 1000-4000 meters below the surface (Pietsch, 2009; Townsend, 2012). In the twilight zone, light sensing organs are the predominant form of communication and organisms show an incredible diversity of light-organ systems. In fact, 95% of mesopelagic fishes have bioluminescent organs of some kind (Pietsch, 2008; Townsend 2012). M. johnsonii has been collected here from the depths of 390-2000 meters below the surface . In the bathypelagic zone the only light produced is from the deep-sea anglerfish lures. M. murrayi is the deepest dwelling species of the Melanocetus found 1000-2500 meters below the surface (Pietsch, 2009). The lack of light in this zone means that visual systems are not as useful an adaptation as they are in the mesopelagic zone. In fact, in the food poor region of the bathypelagic, eyes are a high metabolic burden (Pietsch, 2008).
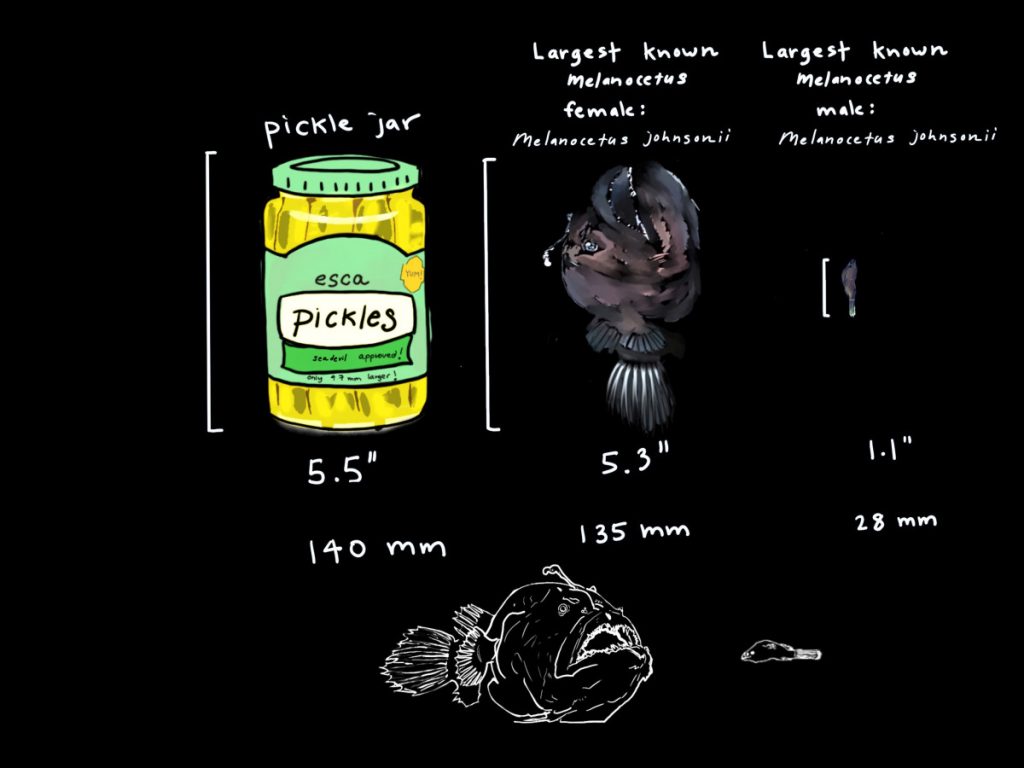
The females of Melanocetidae are short-bodied globular forms with large pendulous bellies that are capable of greatly distending. This form means they can engulf prey considerably larger than themselves, but this form does not make for fast locomotion (Pietsch, 2009). The females globular forms may not be speedy, but they are perfectly developed for hunting and hunting prey considerably larger than themselves. Their large, well toothed mouth is dorsally directed and almost vertical in this family of fishes. When freshly caught the skin is a dark brown velvety black (Pietsch, 2009), which would help to camouflage the species in the depths of the bathypelagic.
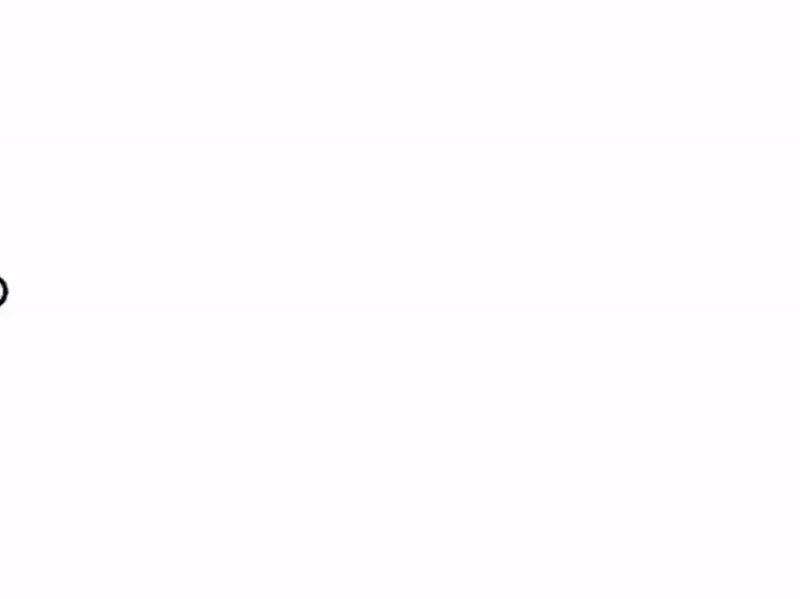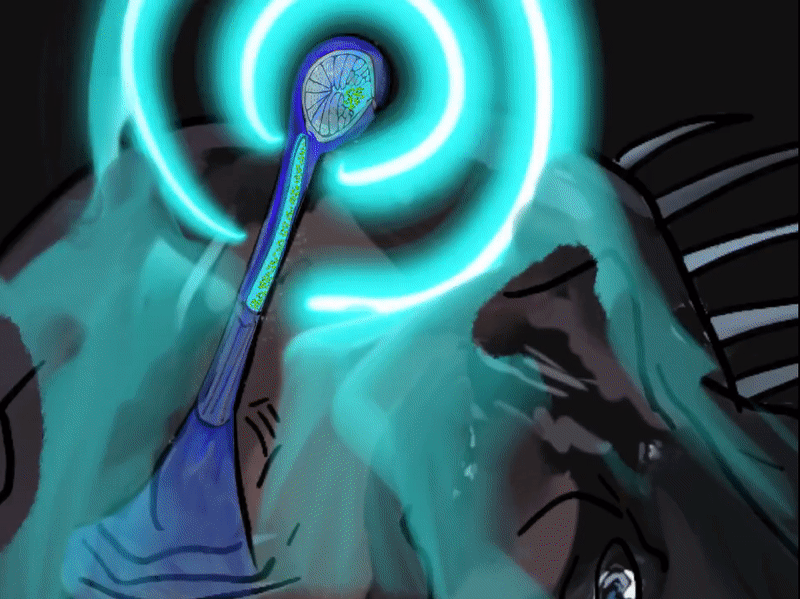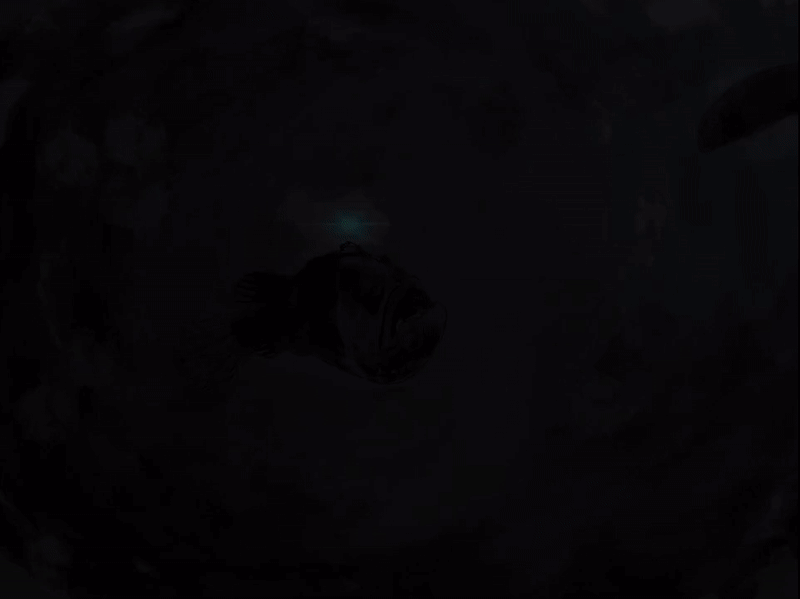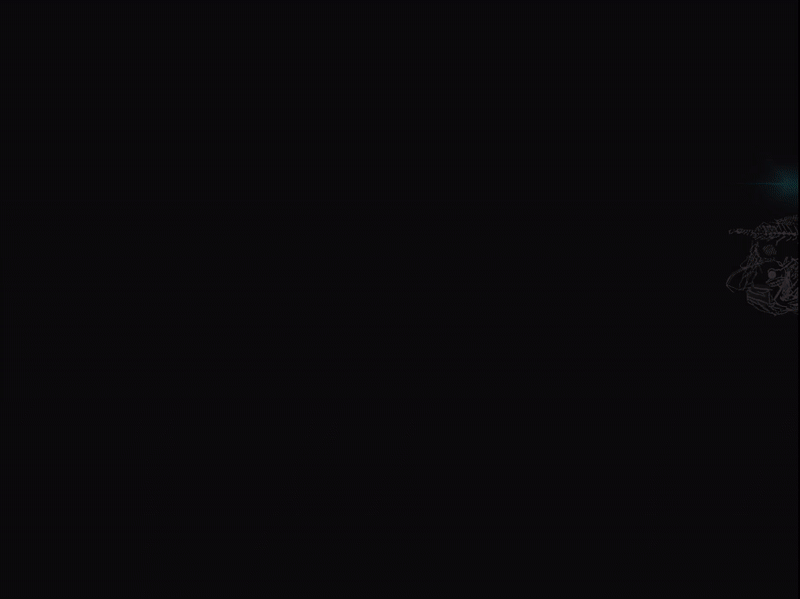
The illicit is a modified first dorsal fin spine that sits on the head of the Melanocetus, it bears the esca (Pietsch, 2009; Townsend, 2012). The esca, Latin for “bait”, is also referred to as the escal or escal bulb. It is thought to be an adaptation for luring in prey, and compared to other members of the seadevil suborder,Ceratioidei, the black seadevil’s esca takes on a simple form without elongated filaments or appendages (Pietsch, 2009). This photophore, photos—Greek for “light” and phoros “a bearing”, consists of a heavily pigmented outer layer, an inner silvery reflective layer and a core and tip of transparent tissue (Pietsch, 2009). A symbiotic relationship with luminous bacteria symbionts are what give the Melanocetus photophore its light. The bacteria are housed in numerous tubular glands that open up into a central cavity (Pietsch, 2009). The mechanisms by which light is generated and controlled is not yet fully understood. The elastic distal end of the escal and a cavity on the side of the bulb are stretched allowing passage of light. Pietsch and Van Duzer (1980) observed in a Melanocetus fish that light emitted “appeared to be controlled by an up and down movement of the darkly pigmented, inner wall of the photophore” (p.241), allowing the fish to rapidly extinguish light (Pietsch, 2009). The color of light produced by the esca is probably due to its passage through modified cells (Pietsch, 2009). In Ceratioide taxa light emitted has been recorded peaking at 470-490 nm which corresponds to the range emitted by most luminous bacteria at 475-510 nm (Pietsch, 2009). All different ranges of light have been recorded with blue/green luminescence transmitting the furthest in oceanic water and the one that deep-sea animals’s eyes are the most sensitive to (Pietsch, 2009).

The luminous bacteria of the Ceratioid are rod like in shape and gram negative. The symbiont bacteria group with, but are not phylogenetically closely related to the P. Phosporeum. They represent a unique lineage related to free-living marine bacteria genus, Vibrio, and the bacteria appear to be species specific throughout the suborder (Pietsch, 2009). It is hypothesized that the symbiont bacteria is free floating and environmentally acquired–based on the retention of genes used for cell wall synthesis and locomotion (Baker et al., 2019). The bacteria are thought to gain access to the light gland through the escal pore and duct (Haygood, 1992; Pietsch, 2009). It was discovered based on their genome that the luminous bacteria of anglerfish have genetically reduced and don’t have the metabolic capacity to use carbon sources that would be readily available in their environment and so they are thought to be obligately dependent on their host (Baker et al, 2019, Hansell 2013, Hendry et.al, 2018 ). This is an exciting find as it represents a possible new form of symbiotic relationship between symbiont and host, one previously unknown to science. In other marine organisms and bioluminescent symbiotic relationships if the bacteria has genetically reduced to depend on the organism they live inside the body (Baker et al., 2019; Pietsch 2009). For a bacteria to genetically reduce but continue to spend the life cycle independently is new and something that is in need of further study!

UMWELT OF THE BLACK SEADEVIL
The life of a female anglerfish may seem solitary, moving through the gloom their globular bodies bounce through the darkness. But they are not alone. The lights from their escal ping into the darkness, pulsating slowly. Inside this beautiful photophore a microscopic world teems with life. Together the Melanocetus and bacteria live in symbiotic harmony. The do not seek out the world, but instead let the world come to them. The bacteria thriving thanks to a willing host, and the beautiful but deadly light attracts dinner for both.
Author: Margaret Lezcano
All artwork by Margaret Lezcano copyrighted © 2020
WORKS CITED
Baker, L. J., Freed, L. L., Easson, C. G., Lopez, J. V., Fenolio, D., Sutton, T. T., Nyholm, S. V., & Hendry, T. A. (2019). Diverse deep-sea anglerfishes share a genetically reduced luminous symbiont that is acquired from the environment. eLife, (8), e47606. https://doi.org/10.7554/eLife.47606
Hansell, D.A. (2013). Recalcitrant dissolved organic carbon fractions. Annual Review of Marine Science (5), 421–445.https://doi.org/10.1146/annurev-marine-120710-100757
Haygood, & M.G., Distel DL. (1993). Bioluminescent symbionts of flashlight fishes and deep-sea anglerfishes form unique lineages related to the genus Vibrio. Nature. (363), 154–156. https://doi: 10.1038/363154a0.
Hendry, T.A., Freed, L.L, Fader, D., Fenolio, D., Sutton, T.T., & Lopez, J.V. (2018). Transposon-mediated genome reduction in the luminous bacterial symbionts of deep-sea ceratioid anglerfishes. mBio. (9), e01033. https://doi.org/10.1128/mBio.01033-18
Pietsch, T.W. (2008). Biology of Fishes. University of Washington.
Pietsch, T.W. (2009). Oceanic Anglerfishes: Extraordinary Diversity in the Deep Sea. University of California Press.
Pietsch, T.W., & J.P. Van Duzer. (1980). Systematics and
distribution of ceratioid anglerfishes of the family
Melanocetidae, with description of a new species from the
eastern North Pacific Ocean. U.S. Fish. Bull., 78(1), 59–87. https://spo.nmfs.noaa.gov/fb.htm
Townsend, D. (2012). Oceanography and Marine Biology: An Introduction to Marine Science. Sinauer Associates Publishing.

