Author – Chelsea Rabourn
Common name: Sea Gooseberry
Pleurobrachia bachei is a gelatinous, tentaculate Ctenophore found in the epipelagic (surface) waters of the Pacific NW coast, from southeast Alaska to Mexico. Pleurobrachia bachei’s distribution tends to be within 5 km of the shore and can reach 50 m deep, “living mostly in the upper 15 m in the day and at about 30 m at night.” (Hirota, 1974)

The phylum Ctenophora, to which Pleurobrachia bachei belongs, are a group of jellyfish look-alikes known as “comb jellies”. Though their name associates these creatures with jellyfish, jellies belong to the phylum Cnidaria and are thus evolution-wise an entirely different type of organism than comb jellies. Ctenophora and Cnidaria both possess bodies comprised of layers of jelly, but in Ctenophores these jelly-body layers are two cells deep and in Cnidaria they are one cell deep.
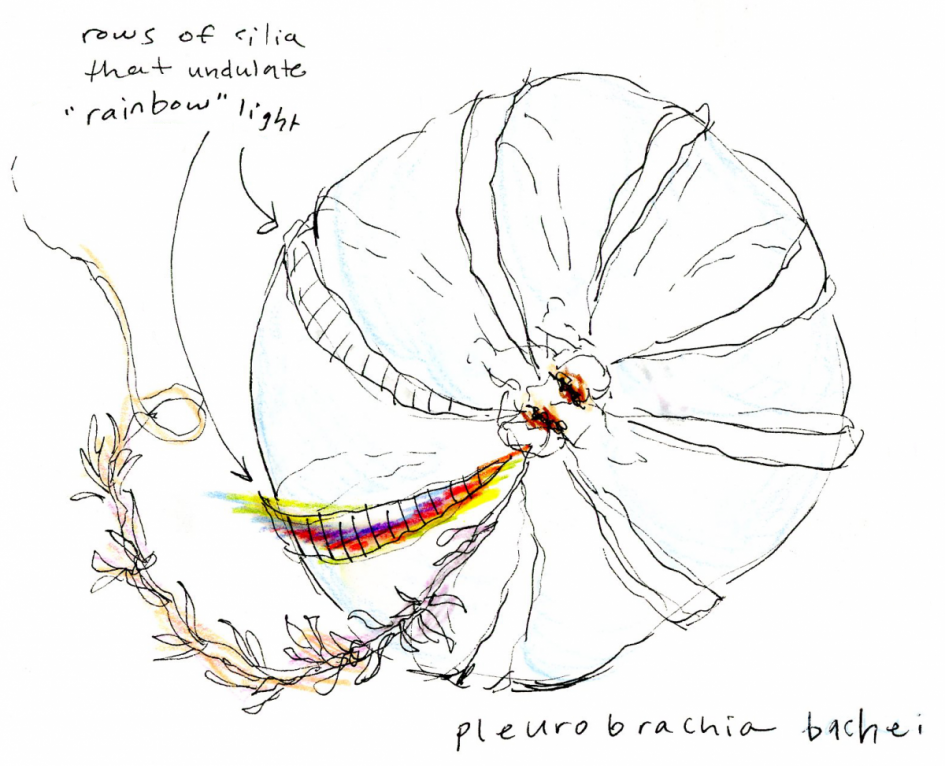
Comb jellies are named for the many rows of comb plates that line their bodies. In Greek, Ctenophore means “comb-bearing”. Each comb row is comprised of thousands of cilia, which oscillate at rapid speeds and allow the Ctenophore to locomote. Fun fact: Ctenophores are the largest non-colonial animals that move primarily through the use of their cilia. As the cilia oscillate they refract light, which to the human eye creates a mesmerizing, colorful display similar to a rainbow. To the untrained eye this beautiful display gives a false appearance of bio-luminescence. Some comb jellies do in fact produce bio-luminescence, but this is a separate process than the refraction of light on their combs, and Pleurobrachia bachei is not a bio-luminescent species.
Confusingly enough Ctenophora and Cnidaria also exhibit some predation behavior that looks similar but is in fact different. Cnidaria like jellyfish and sea anemones are popularly known for their cnidocytes, or explosive cells. An organism possessing cnidocytes fires a venomous structure from a subcellular organelle called a nematocyst upon contact with their prey. This action produces a sometimes painful and often paralyzing sting to the prey of Cnidaria. In other words, Cnidaria possess “stinging cells”. Jellyfish are passive carnivores; nematocysts line the long tentacles that flow from their Medusa form and catch their prey by stinging prey organisms upon contact. Comb jellies are also passive carnivores and possess tentacles for catching their prey. However, comb jelly tentacles do not possess cnidocytes or nematocysts. Instead they possess colloblasts, which shoot an adhesive substance rather than a venomous sting into their prey. Once a prey organism adheres to the tentacle of a Ctenophore, the comb jelly will pull the tentacle back to its body and consume its prey.
When searching for prey, P. bachei releases its two long tentacles and twirls its body until the tentacles unfurl and become free to flow behind and around its globular body. That achieved, P. bachei will passively drift until prey becomes stuck in their tentacles via the adhesive substance emitted by their colloblasts. When P. bachei is ready to eat, its tentacles will be slowly retracted toward its oral end for consumption. (Greene et al., 1986)
When fed copepods and other zooplankton from Puget Sound, P. bachei demonstrated that it does not simply consume whatever is caught in its tentacles, despite its passive feeding technique. “Pleurobrachia [bachei] is a selective carnivore, and its diet is determined by both relative availability and vulnerability of prey.” P. bachei selectively consumed zooplankton at different life stages when prey were transitioning from one life stage to another and were thus more vulnerable to easy handling and consumption by the Ctenophore. This discovery contradicted previous assumptions that Pleurobrachia bachei was a non-selective passive carnivore. Instead, P. bachei is now understood to be a selective ambush-predator that takes advantage of prey in different stages of vulnerability. (Greene et al., 1986)
Pleurobrachia bachei’s symbolic message to us is one of patience. As this comb jelly waits for its prey to be caught in its tentacles, we are reminded to let life come to us. In times of crisis such as the present, when anthropogenic climate change threatens the health of fellow species and humankind, it is easy to get ahead of ourselves and rush to action before we know what the next right step may be. Sometimes the best response to crisis is a passive absorption of the blow, followed by a discerning choice at the appropriate moment. When prey is caught in P. bachei’s tentacles it does not immediately consume the prey; it discerns the vulnerability of its prey and any danger a particular prey species may pose to P. bachei’s body. Only when P. bachei senses that their prey is safe to eat and nutritious do they choose to finally consume their catch. We would be wise to respond to crises with the patience and discernment Pleurobrachia bachei models.

Literature Cited
Hirota, J. (1974) “Quantitative natural history of Pleurobrachia bachei in La Jolla Bight.” Fish. Bull 72.2: 295-335.
Greene, Charles H., Landry Michael R., and Monger Bruce C. (1986) “Foraging Behavior and Prey Selection by the Ambush Entangling Predator Pleurobrachia Bachei.” Ecology 67.6: 1493-501. Web.